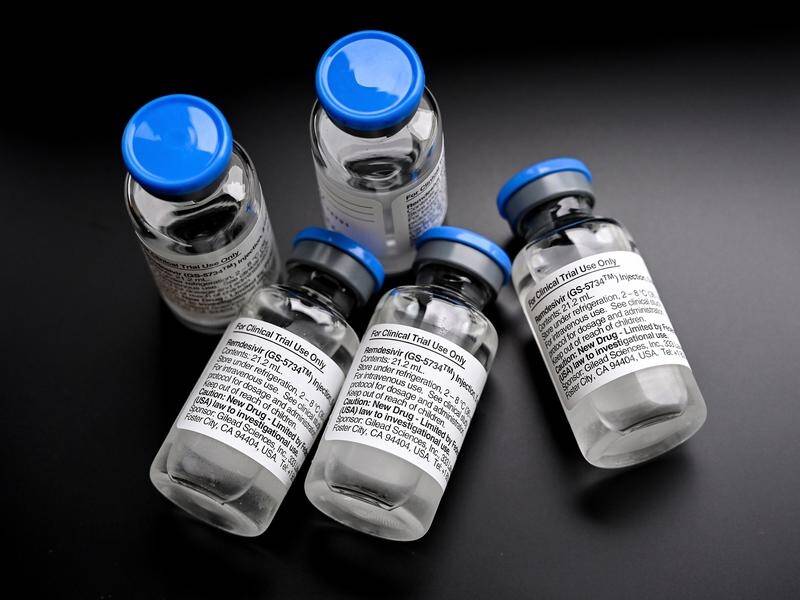
The Therapeutic Goods Administration has given provisional approval to a drug that can improve the recovery time of the most seriously ill COVID-19 patients.
Subscribe now for unlimited access.
$0/
(min cost $0)
or signup to continue reading
The approval for remdesivir comes as Australia sees a sharp increase in coronavirus cases in Victoria, which has sparked fears the outbreak may spread across the country.
Remdesivir has been provisionally approved for use in adults and adolescent patients with severe COVID-19 symptoms who have been hospitalised.
The drug by US company Gilead Sciences is the most promising treatment so far to reduce hospitalisation time for those suffering from severe coronavirus infections, the TGA said in a statement on Friday.
The TGA says remdesivir will be available only to patients who are severely unwell, require oxygen or high level support to breathe, and are in hospital care.
However, the drug does not prevent coronavirus infection or relieve milder cases.
Australia is one of the first countries to approve the use of the drug after the European Union, Japan, and Singapore.
Australian Associated Press

